| STORY CREDITS Writer: Devki Prabhu Editor: Prof Krishna Kanti Dey |
Study reveals how enzyme activity can influence surrounding particles in a crowded environment, reshaping our understanding of intracellular motion.
Complexity is what truly defines the human body with its intricate network of biochemical pathways, at the heart of which, are enzymes—proteins that catalyse essential life-sustaining reactions. Much like a toaster that transforms a slice of bread into toast, enzymes convert their substrates into products that power vital cellular functions.
In a recent study, researchers at the Indian Institute of Technology Gandhinagar (IITGN) have uncovered new insights into how enzymes influence their surroundings beyond their chemical roles. The work, titled “Propagation of Enzyme-driven Active Fluctuations in Crowded Milieu,” published in Small (Wiley-VCH), reveals how enzymes can transmit mechanical energy in dense, cell-like environments through subtle acoustic-like waves. Previous research shows that this energy aids in the movement of surrounding particles called ‘tracers’ in water-based environments.
This study, led by Prof Krishna K Dey, Associate Professor of Physics at IITGN, elucidates that in spite of the viscosity of the media that were used to mimic the crowded interiors of cells, enzymes such as catalase and urease can enhance the motion of nearby microscopic tracer particles. This enhancement occurred even with lower reaction rates compared to simple water-based environments — a surprising finding that challenges conventional understanding of molecular motion and energy transfer in crowded intracellular environments. “It is important to determine whether enzyme-induced motion can overcome the dissipative effects of crowded intracellular environments,” explained Professor Dey.
The team found that even in identical reaction conditions, the tracers’ motion was most pronounced in crowded media. “This interesting observation prompted us to explore how the intervening media itself influences energy transfer from the enzymes to tracers,” explained Prof K R Jayaprakash, a co-author and Assistant Professor of the Department of Mechanical Engineering at IITGN.
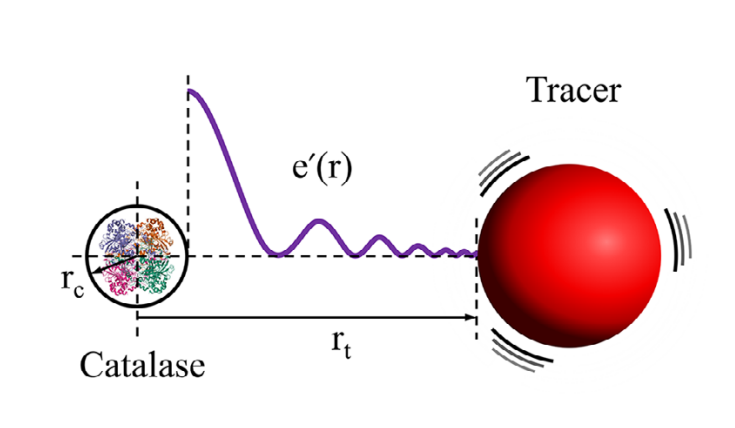
Traditionally, motion in fluids has always been perceived through the lens of hydrodynamics, but the present observations deviate from what is typically expected in water-based environments, which could mean that enzyme-induced motion may involve additional, non-hydrodynamic pathways. “To explain this phenomenon, we have proposed a new alternative mechanism that could possibly explain how these tracers’ movements are being controlled in crowded environments,” explained Rik Chakraborty, the first author of the study.
The team developed an “Activity Induced Acoustic Stress Model,” which suggests that during catalysis, enzymes generate transient mechanical pressure waves that propagate through the surrounding medium. These waves can transmit energy to nearby particles, enhancing their motion even in viscous, cell-like conditions. Analytical calculations supported the experimental findings, indicating that the surrounding medium plays a crucial role in transmitting sound-like fluctuations, and that multiple mechanisms may govern molecular motion in complex biological environments.
“We believe these findings can potentially reshape such enzyme-driven designs for micro- and nanomotors used for minimally invasive surgeries, targeted drug delivery, and other biomedical applications,” added Diptangshu Paul, a co-author from the Department of Mechanical Engineering. Reflecting on the broader significance of the work, Arnab Maiti, another co-author, remarked that the study highlights how small molecular players, like enzymes, can exert far-reaching effects beyond enzymology.
“In today’s world, it is important to understand the fundamentals of any concept to be able to apply the outcomes effectively,” noted Chakraborty. “This is one such study that tells us a better story of how an enzyme and its behaviour toward its surroundings can be applied in this era of technology for human benefit.”
The research was supported by Anusandhan National Research Foundation (ANRF), Ministry of Education (MoE-STARS), and the Gujarat State Biotechnology Mission (GSBTM).